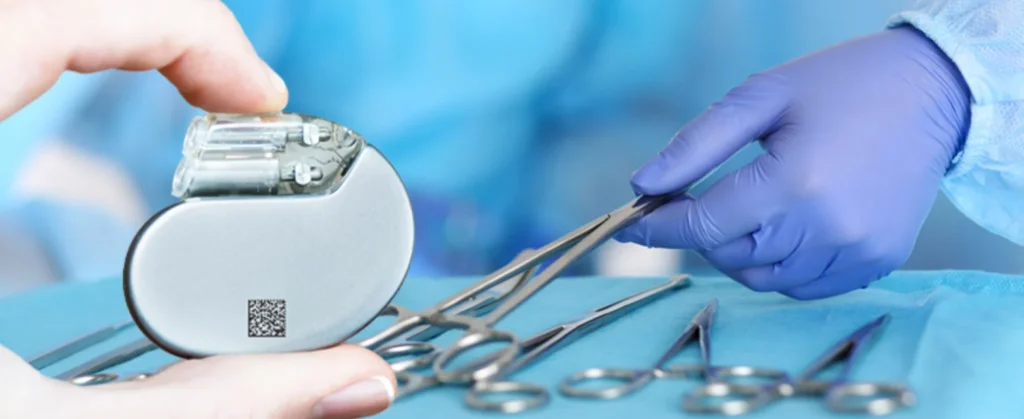
Medical devices are subject to strict marking regulations in the European Union and the USA. In both regions, medical products and devices have to be marked with a UDI. UDI stands for Unique Device Identifier. In the EU, the exact rules for UDI marking are defined in the Medical Device Regulation (MDR) 2017/745. Since 2021, manufacturers, distributors and importers of medical devices have to implement this unique device identifier in stages. By 2027, medical devices of all hazard classes and their packaging will have to bear the UDI marking as a 1D Code or 2D Code and in plain text. This is intended to further improve traceability, counterfeit protection, product safety and ultimately patient safety.
Whether hip prostheses or dental implants, surgical instruments or insulin pumps, bandages or care beds: the marking technology used to ensure that the UDI and other markings on medical devices are easily accessible and permanent depends on the material and the specific application of the medical device.
Challenges with markings for medical technology
- Very strict normative labeling requirements (MDR 2017/745)
- Ensuring optimal readability and contrast on a permanent basis
- A lot of information on sometimes very small areas
- Ensuring forgery-proof and patient safety
- Serialization / Track & Trace